Booster and Additional Primary Dose COVID-19 Vaccinations Among Adults Aged ≥65 Years — United States, August 13, 2021–November 19, 2021 | MMWR
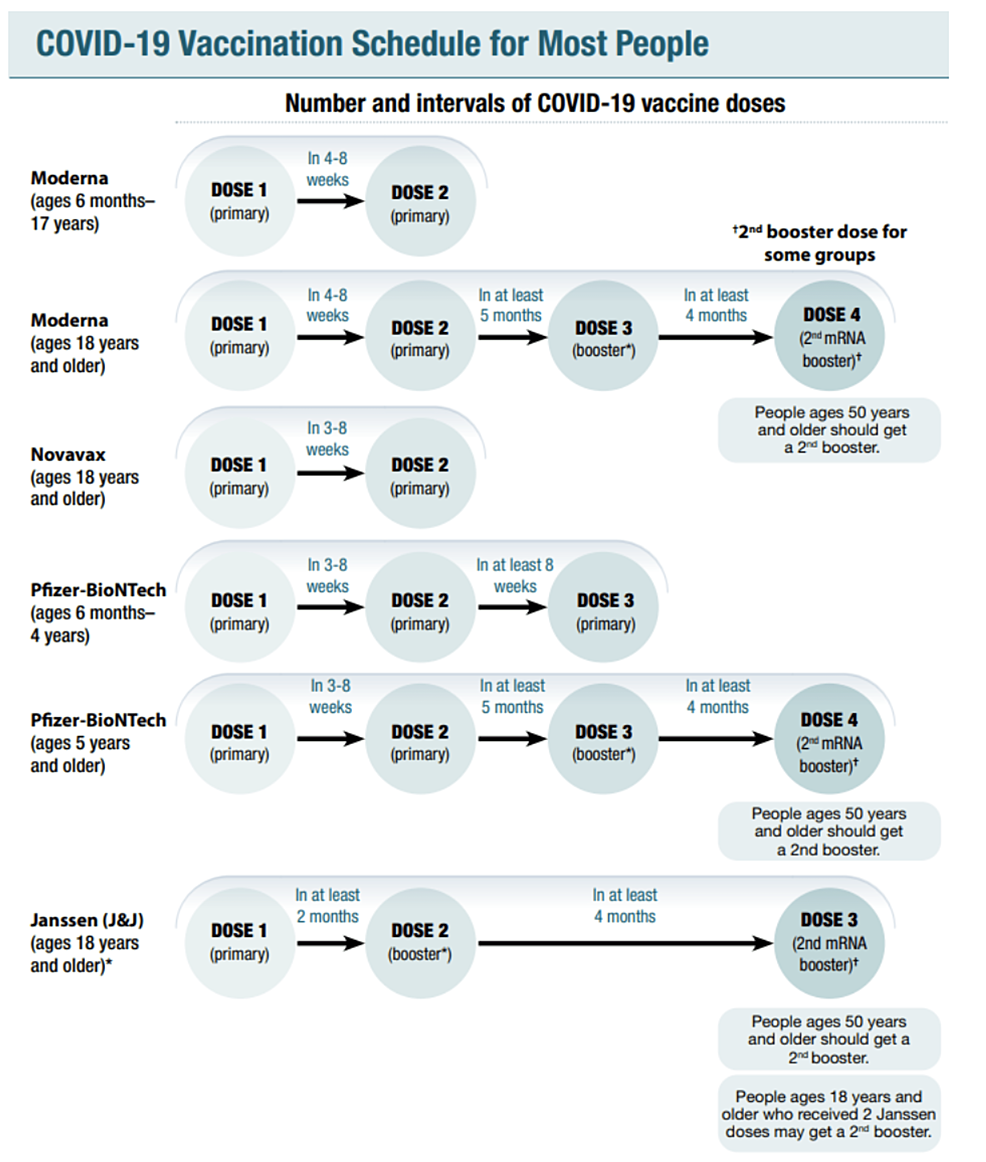
Cureus | A Hitchhiker's Guide to Worldwide COVID-19 Vaccinations: A Detailed Review of Monovalent and Bivalent Vaccine Schedules, COVID-19 Vaccine Side Effects, and Effectiveness Against Omicron and Delta Variants | Article

Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults — Increasing Community Access to Testing ...

Frontiers | Association Between Risk Perception and Acceptance for a Booster Dose of COVID-19 Vaccine to Children Among Child Caregivers in China
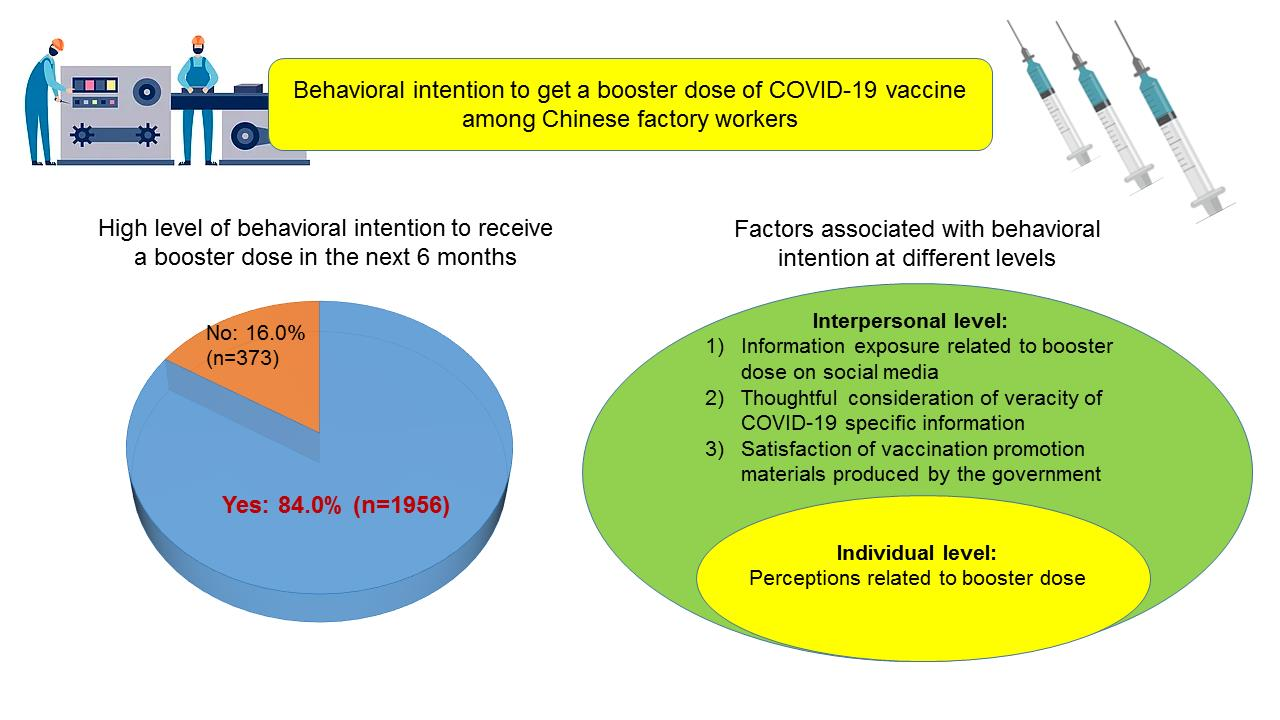
IJERPH | Free Full-Text | Behavioral Intention to Get a Booster Dose of COVID-19 Vaccine among Chinese Factory Workers

CDC on X: "The definition of fully vaccinated has not changed with expanded use of booster doses. You're considered fully vaccinated against #COVID19: - 2 weeks after your second dose of a

MenACWY-CRM conjugate vaccine booster dose given 4–6 years after priming: Results from a phase IIIb, multicenter, open label study in adolescents and adults - ScienceDirect

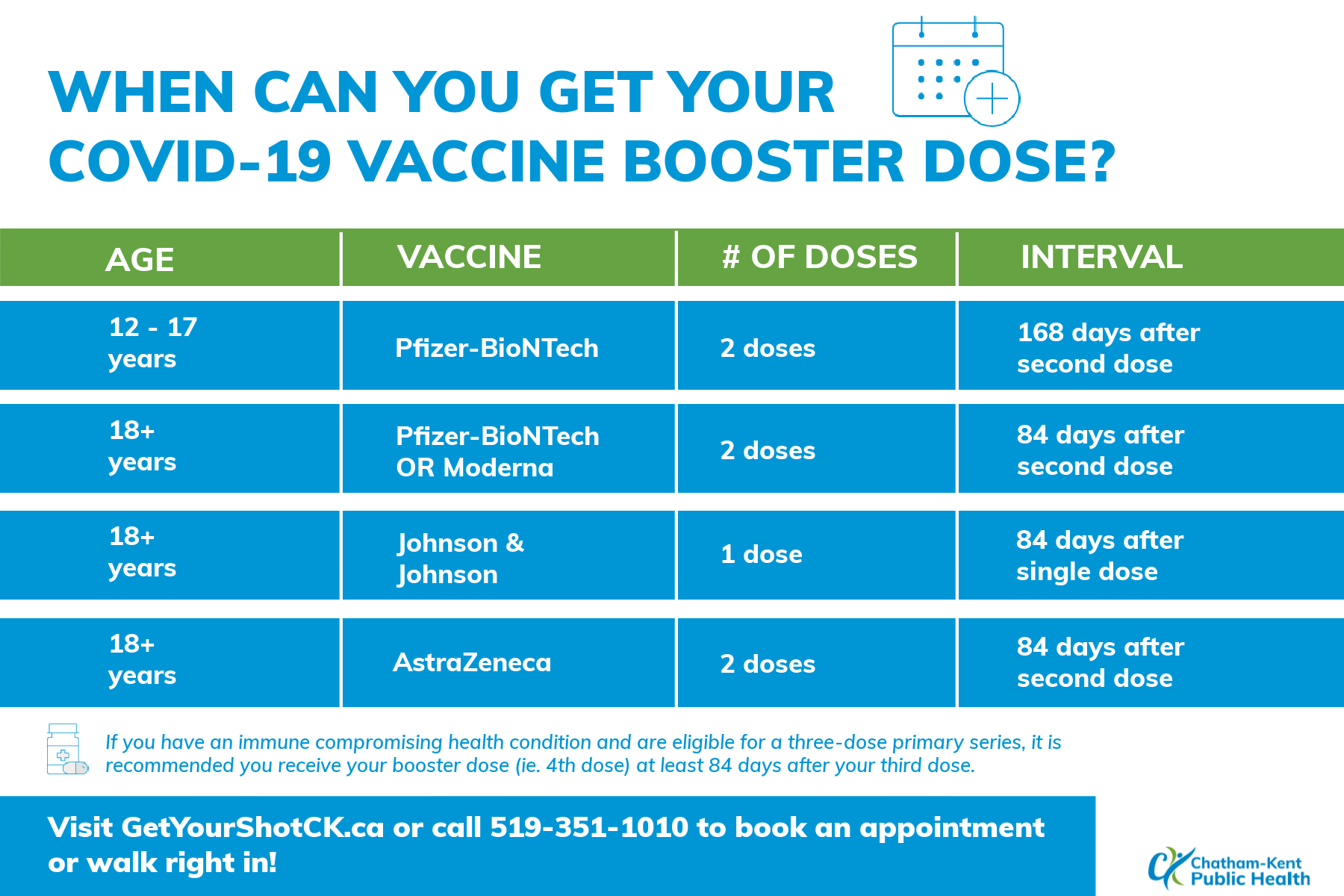
Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA

CDC - After FDA approves and ACIP recommends a COVID-19 booster dose, the goal is for those who received their shots in the earlier phases of vaccine rollout to get their booster

Coronavirus (COVID-19) Update: FDA Takes Multiple Actions to Expand Use of Pfizer-BioNTech COVID-19 Vaccine | FDA